UT-Austin Proof of Concept Study
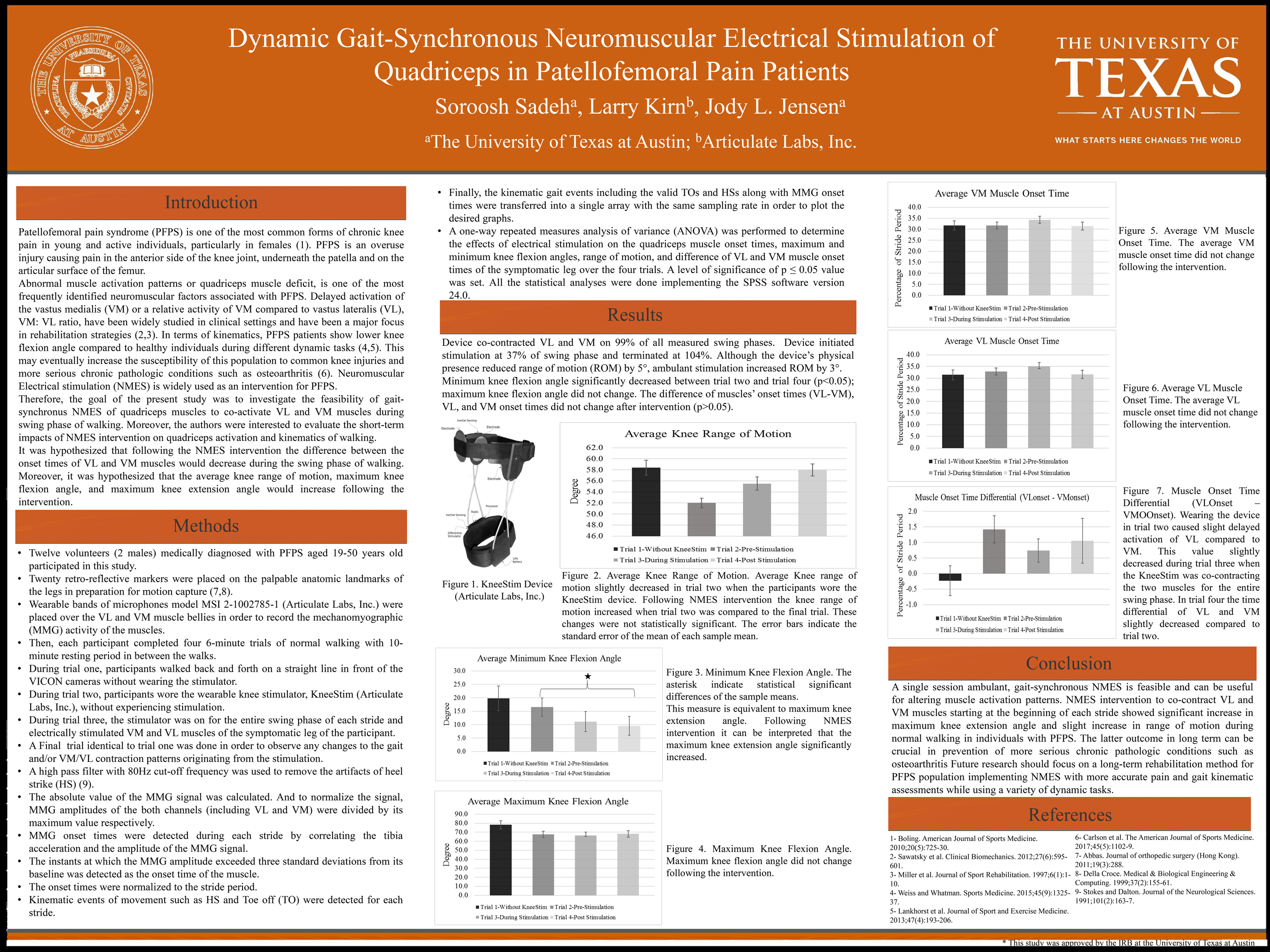
Research Objectives
To investigate the feasibility of ambulant, gait-synchronous neuromuscular electrical stimulation (NMES) during swing phase and evaluate short-term impacts on quadriceps activation and walking kinematics to potentially treat Patellofemoral pain syndrome (PFPS).
Design
Prospective, controlled, open label, single-center study where participants will have their gait and quadriceps muscle activation patterns evaluated through four six-minute walk tests (W1 through W4) with or without the electrical stimulator device being worn and with or without the device being operational.
Setting
Testing occurred at UT-Austin’s Developmental Control Laboratory with a 10-camera VICON system and mechanomyographic (MMG) sensors.
Participants
12 subjects with evidence of PFPS as measured by Anterior Knee Pain Scale scores and patella glide, tilt, and grind tests.
Interventions
- A novel NMES device was designed to enable gait-synchronous muscle stimulation.
- For PFPS subjects, device was instructed to co-contract both vastus laterallis (VL) and vastus medallis (VM) during swing phase to address potential contraction timing disparity that may exacerbate PFPS.
Main Outcome Measures
- MMG readings of VL and VM
- Kinematics of movement recorded by VICON motion system
Results
Device co-contracted VL and VM on 99% of all measured swing phases. Device initiated stimulation at 37% of swing phase and terminated at 104%. Although the device’s physical presence reduced range of motion (ROM) by 5°, ambulant stimulation increased ROM by 3°.
Maximum knee extension angle (xx) significantly increased between W2 and W4; maximum knee flexion angle (1.64%) did not. The device altered VL/VM contraction onset times from W2 to W3 (6.8%, 10.2% respectively) but changes did not persist through W4.
Conclusions
Ambulant, gait-synchronous NMES is feasible and may be useful for altering muscle activation patterns. Further research is required to evaluate long-term implications.
Author Disclosures
Articulate Labs, Inc, sponsored this study.
View the Full Study at the Archives of Physical Medicine and Rehabilitation
